Table of Contents
n
- n
- What Are Fluorescence Filter Sets and How Do They Work?
- Matching Filters to Fluorophores: Overlap, Stokes Shift, and Bandwidths
- Light Sources: LEDs, Lasers, and Arc Lamps for Fluorescence Microscopy
- Filter Technology, Optical Density, and Angle Sensitivity
- Practical Setup and Alignment of Filter Cubes and Fluorescence Paths
- Avoiding Bleed-Through, Autofluorescence, and Background
- Multicolor Imaging Strategies: Single-Band, Multiband, and Spectral Unmixing Accessories
- Compatibility, Mechanical Considerations, and Cube Formats
- Frequently Asked Questions
- Final Thoughts on Choosing the Right Fluorescence Filters and Light Source
n
n
n
n
n
n
n
n
n
n
nn
What Are Fluorescence Filter Sets and How Do They Work?
n
Fluorescence microscopy relies on a specific arrangement of optical filters and mirrors to selectively excite fluorescent molecules (fluorophores) and collect their emitted light while strongly rejecting unwanted wavelengths. The core accessory that enables this is the fluorescence filter set, often packaged inside a filter cube mounted in the microscope’s epi-fluorescence turret. Understanding the role of each component is the foundation for selecting and using the right accessories for clear, high-contrast images.
nn
 n
nn
nn
A typical fluorescence filter set contains three elements:
n
- n
- Excitation filter – Passes only the wavelengths needed to excite the fluorophore, blocking other parts of the spectrum from the light source.
- Dichroic beamsplitter (dichroic mirror) – A wavelength-selective mirror placed at 45°. It reflects the excitation band down through the objective onto the sample and transmits the longer-wavelength fluorescence emission back up to the detector.
- Emission filter – Also known as a barrier filter, it passes the fluorophore’s emitted light while blocking residual excitation and other stray wavelengths before the detector (eyes or camera).
n
n
n
n
Physically, in an epi-fluorescence configuration, the light source is directed through the excitation filter, hits the dichroic at 45°, and is reflected into the objective lens and onto the specimen. Fluorescence generated by the specimen travels back through the objective, meets the dichroic again, and this time the longer-wavelength emission is transmitted through the dichroic, then through the emission filter to the detector. The dichroic’s ability to reflect shorter wavelengths and transmit longer ones underpins the optical separation of excitation and emission. This separation is necessary because the excitation light is typically far brighter than the fluorescence signal, and even small leaks can overwhelm the image.
n
Fluorescence filter sets are usually defined by their spectral passbands. You may see sets labeled as “FITC,” “TRITC,” “DAPI,” “Cy3,” “Cy5,” or “GFP,” indicating their target fluorophores, or by numerical passband specifications (for example, excitation and emission bandpass ranges). While labels are helpful, always verify the actual spectral curves because fluorophore variants and imaging conditions can differ. Later sections—especially Matching Filters to Fluorophores and Avoiding Bleed-Through, Autofluorescence, and Background—explain how exact passbands and blocking levels influence image quality.
n
There are several important performance characteristics to understand:
n
- n
- Bandpass shape and width – A bandpass filter transmits a defined wavelength range and blocks others. Narrower bands improve selectivity (useful for reducing background and bleed-through), while wider bands can admit more light (useful for brighter signals). Longpass and shortpass filters transmit all wavelengths longer or shorter than a cutoff, respectively.
- Optical density (OD) blocking – Outside the passband, a filter’s blocking is specified by OD, describing how strongly it attenuates light. It is standard to require high OD blocking at the excitation wavelength in the emission channel to suppress leakage.
- Surface quality and flatness – To avoid introducing aberrations or stray reflections, filters are polished and coated to high standards. Flatness and parallelism matter more as objectives become more demanding and cameras more sensitive.
- Angle sensitivity – Interference filters shift their passbands slightly when tilted or when used with converging or diverging beams. This is especially relevant in epi-fluorescence where the light cone through the objective spans a range of angles. We’ll expand on this under Filter Technology, Optical Density, and Angle Sensitivity.
n
n
n
n
n
Finally, note that a fluorescence cube is integrated into a broader optical system: the light source and its optics, the microscope’s illumination and detection paths, and the detector (eyepieces or a camera). A well-matched set aligns the filter passbands to the fluorophore and the light source spectrum. Even the best filters cannot compensate for significant mismatch in illumination (for example, trying to excite a fluorophore whose main absorption band is underpowered by the source). To choose effectively, it helps to consider filters and sources together, as we do in Light Sources: LEDs, Lasers, and Arc Lamps and then connect those insights back to Matching Filters to Fluorophores.
nn
Matching Filters to Fluorophores: Overlap, Stokes Shift, and Bandwidths
nn
 n
nn
nn
Selecting a filter set starts with the fluorophore’s spectra. Most fluorophores have:
n
- n
- Excitation spectrum – The efficiency of fluorescence emission as a function of excitation wavelength.
- Emission spectrum – The distribution of emitted light intensities over wavelength after excitation.
n
n
n
The difference between the excitation and emission peak wavelengths is the Stokes shift. Larger Stokes shifts simplify filter design because they help ensure that the emission passes through the detection path while excitation is strongly rejected. Smaller Stokes shifts require tighter control over filter passbands and higher blocking outside the bands to avoid contamination from the much brighter excitation light.
n
Here is a practical framework for selecting filters for a single fluorophore:
n
- n
- Locate the excitation peak and choose an excitation filter that passes a region of high excitation efficiency while rejecting wavelengths that would unnecessarily increase background. When possible, align the passband to strong emission from the light source (see Light Sources), maximizing signal without bleeding into the emission range.
- Choose the dichroic cutoff so that it reflects the excitation band effectively and transmits the emission band with minimal loss. The cutoff wavelength is the transition region; its slope should be steep to minimize the band of overlap.
- Select the emission filter to pass the strongest and cleanest part of the emission spectrum while blocking the excitation band and other out-of-band wavelengths. Wider emission bands can boost signal, but if the excitation and emission are close, a narrower emission band often yields higher contrast.
n
n
n
n
For fluorophores with broad spectra, consider these trade-offs:
n
- n
- Wide bandpass filters (both excitation and emission) capture more light and can reduce exposure times but may increase background or introduce crosstalk when imaging multiple labels.
- Narrow bandpass filters yield higher spectral selectivity, reducing background and bleed-through, but they can diminish signal and lengthen exposures.
- Longpass emission filters are attractive for dyes with long, smooth emission tails, but if the excitation band lies close to the emission, longpass designs can admit stray excitation leakage unless blocking is very high.
n
n
n
n
For multicolor imaging, the main challenges are spectral crosstalk and sequential acquisition strategy. If two fluorophores have overlapping spectra, tightening the passbands or switching to single-band filters per channel and imaging them one at a time often reduces crosstalk. Multi-band filter sets can simplify workflows and switching times, but they require careful selection to avoid confusing signal channels. We unpack these approaches in Multicolor Imaging Strategies.
n
In all cases, check the following:
n
- n
- Excitation–emission separation – Inspect the manufacturer’s spectral plots for the filter set. There should be a well-defined gap between excitation and emission passbands with high OD blocking in the gap at relevant wavelengths.
- Source matching – The excitation filter should transmit wavelengths where your light source is strong. For example, if your LED source peaks at a particular line, align the passband accordingly. If using a laser line, the excitation filter should admit that line with minimal loss and block nearby lines that are not needed.
- Photophysics of the fluorophore – Some dyes bleach faster under certain excitation bands. If the fluorophore’s excitation spectrum has multiple lobes, it can be helpful to select a band that gives sufficient signal while reducing photobleaching risk.
- Environmental factors – pH, solvent, and binding state can shift spectra slightly for some fluorophores. When possible, choose filter sets with a bit of margin in passbands to accommodate small spectral shifts.
n
n
n
n
n
Example thought process (no specific brands required): imagine choosing a filter set for a green fluorescent protein with excitation near the blue–cyan range and emission in green. You would pick an excitation filter centered near the protein’s strong absorption region (commonly in the blue), a dichroic that reflects that blue band and transmits green, and an emission filter centered on the green emission with high blocking back into the blue. If you later decide to add a red-emitting dye to the sample, you would ensure that the green channel’s emission filter does not pass significant red light and that the red channel’s excitation does not overlap the green emission band. If overlap is unavoidable, sequential imaging and tighter filters can help; see Avoiding Bleed-Through, Autofluorescence, and Background for detailed strategies.
n
Finally, it’s useful to familiarize yourself with some standard spectral terms common in datasheets:
n
- n
- CW (center wavelength) – Midpoint of the passband.
- FWHM (full width at half maximum) – Bandwidth where transmission is at least half of its peak value.
- Peak transmission – The maximum throughput inside the passband, ideally high (often above 90% for modern hard-coated filters).
- Blocking range – The wavelength range over which the filter provides a specified minimum OD. Effective rejection across this range is crucial in fluorescence detection because any out-of-band light competes with the relatively weak emission signal.
n
n
n
n
nn
Light Sources: LEDs, Lasers, and Arc Lamps for Fluorescence Microscopy
n
Filters and sources are two sides of the same coin. A great filter set paired with a poorly matched or unstable light source will still produce suboptimal results. Here are the common illumination options for epi-fluorescence microscopy and how to think about their pairing with filters.
n
LED illumination
n
High-power light-emitting diodes have become a dominant choice for widefield fluorescence due to their stability, longevity, and ease of use. Their spectral output is typically concentrated into moderately broad peaks centered near common excitation bands (for example, violet, blue, cyan, green, and red). Advantages include:
n
- n
- Instant on/off and fast modulation – Easy electronic shuttering enables rapid switching between colors, helpful for multiplexed imaging. This also reduces exposure time outside of acquisition, minimizing photobleaching.
- Stable output – Well-designed LED systems warm up quickly and deliver consistent intensity over long sessions, which is beneficial for quantitative imaging.
- Lower maintenance – LEDs do not require bulb replacement at short intervals.
n
n
n
n
When pairing LEDs with filters, ensure the excitation filter passband aligns with the LED’s emission peak. If the LED bandwidth extends toward the emission side, a tighter excitation bandpass or additional order-sorting filter can help block the long-wavelength tail from reaching the sample.
n
Laser illumination
n
Lasers provide near-monochromatic light at defined wavelengths. They are widely used in confocal and advanced widefield systems where precision and high irradiance are required. Their narrow lines simplify excitation filter selection (the filter mainly needs to pass the line and block everything else), but they introduce other considerations:
n
- n
- Speckle and coherence artifacts – Coherent light can produce interference patterns. Spackle mitigation optics (diffusers or fiber coupling with agitation) are often used to improve illumination uniformity.
- Line selection – Because each laser line is fixed, your excitation filter should pass that line with high transmission. For multi-line systems, multiband excitation filters or multiple single-band filters combined with rapid switching may be used.
- Power management – Neutral density filters or electronic modulation are used to adjust intensity without shifting the wavelength.
n
n
n
n
For lasers, match the dichroic cutoff to reflect the laser line(s) while transmitting the emission. Careful attention to the dichroic’s transition slope and blocking near the laser line is essential because even very small leaks can dominate the detected signal given the high intensity of laser illumination.
n
Arc lamps (mercury, xenon, and metal halide)
n
Arc lamps and related sources offer broadband emission with prominent spectral lines. Historically, mercury lamps have been common in fluorescence because their strong line emissions can match common excitation bands. Xenon provides a more continuous spectrum, while metal halide systems combine aspects of both and can couple efficiently into microscopes. Considerations include:
n
- n
- Warm-up and stability – Arc lamps typically require warm-up time for intensity stabilization. Intensity can drift over lamp lifetime, and bulb replacement is periodic.
- Spectral selection – With a broadband source, your excitation filter defines the illumination band. High-quality excitation filters are crucial to avoid passing unwanted lines or tails that might increase background.
- Shuttering and heat – Mechanical shutters may be needed to control exposure. Heat management and proper coupling optics are important to protect optics and samples.
n
n
n
n
In short, LEDs are often the go-to for widefield fluorescence due to stability, ease, and spectral convenience; lasers are excellent for precise excitation and high-intensity applications; and arc/metal-halide sources remain practical, especially where existing systems are in place or strong line excitation is desired. Whichever source you use, its spectrum should be considered alongside your filter set as part of one optically coherent system. If you are balancing multiple fluorophores, consult the Multicolor Imaging Strategies section to ensure your source-switching capabilities and filter passbands support your imaging plan.
nn
Filter Technology, Optical Density, and Angle Sensitivity
n
Most modern fluorescence filters are interference filters composed of multiple dielectric layers. Their performance depends on constructive and destructive interference: within the passband, constructive interference reinforces transmission; outside it, destructive interference leads to attenuation. A few key technical terms are essential for comparing filters:
n
- n
- Optical density (OD) – A logarithmic measure of attenuation outside the passband. It relates to transmission T by the equation:
n
n
OD = -log10(T)nT = 10^(-OD)nnn
 n
nn
nn
Higher OD values correspond to stronger blocking. For example, OD6 corresponds to a transmission of 10^-6 (one part per million). High OD in the emission filter at the excitation wavelength is critical because even small leaks of the bright excitation can outshine the emission signal.
n
- n
- Transition slope – The rapidity with which a filter transitions from stopband to passband. Steeper slopes minimize the region of overlap and allow tighter separation between excitation and emission bands, which is crucial for fluorophores with small Stokes shifts.
- Hard-coated vs. soft-coated – Hard-coated filters employ durable dielectric layers that provide high transmission, robust blocking, and environmental stability. They are generally preferred for high-performance fluorescence microscopy because they minimize aging and delamination issues.
- Surface quality and wavefront error – The physical flatness and polish of the filter substrate affect transmitted and reflected wavefronts. Good surface quality helps maintain image sharpness and reduces ghost images from unintended reflections.
n
n
n
n
Angle sensitivity matters because the effective passband of an interference filter shifts with angle of incidence. When a filter is used at non-zero angles or in a converging beam, the transmitted band tends to shift toward shorter wavelengths (a blue shift). In epi-fluorescence, both the illumination and collection beams pass through the objective lens, which introduces a range of angles defined by the system’s geometry. To cope with this, manufacturers typically design filters and dichroics for the angles encountered in microscope beams. However, if you tilt a filter intentionally (for instance, to fine-tune its passband), be aware of the induced shift and potential degradation of blocking in critical regions.
n
Orientation also matters. Many emission and excitation filters are marked with an orientation arrow indicating the preferred direction of light travel (this can optimize blocking performance and minimize surface reflections). Dichroic mirrors are almost always intended for a specific orientation at 45°, with a coated side facing the incoming excitation beam. Installing these elements correctly is essential for achieving the specified performance; see Practical Setup and Alignment.
n
Lastly, consider stray light control. Internal reflections between flat, parallel surfaces can cause ghost images or background haze. Blackened cube interiors, proper apertures, anti-reflection coatings on ancillary optics, and careful attention to cleanliness help keep stray light low. When in doubt, trace the optical path: if you see a bright region outside the expected field or unexpected flare, it may indicate a reflection from a contaminated surface or an incorrectly oriented filter.
nn
Practical Setup and Alignment of Filter Cubes and Fluorescence Paths
n
A well-chosen filter set only reaches its potential when the illumination and detection paths are aligned, clean, and operating as intended. The following practical considerations focus on optical alignment rather than clinical or laboratory procedures, keeping the guidance educational and system-agnostic.
n
Confirm cube orientation and seating
n
- n
- Inspect each cube to ensure the dichroic and the excitation/emission filters are properly oriented. Look for orientation marks on filters (arrows or text indicating the source side) and the dichroic’s coated face.
- Verify the cube is fully seated in the turret and indexed correctly. Partial insertion can cause vignetting or unexpected reflections.
n
n
n
Check illumination uniformity
n
- n
- With a uniform fluorescent sample or a reference test slide, examine the field illumination. Unevenness can come from source coupling, misalignment of apertures, or contamination in the light path.
- If you see hot spots or gradients, inspect the illumination optics before the cube (collectors, field apertures). In systems with adjustable apertures, center and adjust them following the microscope’s recommendations for even field illumination.
n
n
nn
 n
nn
nn
Control exposure and intensity
n
- n
- Use electronic control when available (LED current, laser modulation) for consistent exposures.
- Employ neutral density (ND) filters to reduce intensity without altering spectral content. ND filters are helpful for bright fluorophores that would otherwise saturate the detector.
n
n
n
Keep optics clean
n
- n
- Dust or residues on filters, dichroics, and nearby lenses can scatter light and raise background. Use appropriate, manufacturer-recommended cleaning methods and avoid scratching coatings.
- Minimize handling—hold filters by the edge and store them in clean, labeled containers when not in use.
n
n
n
Verify detection path integrity
n
- n
- Confirm that the emission filter is correctly positioned in the detection path. Any misplacement can allow excitation leakage.
- Check for unintended polarizing effects or depolarization elements if you are combining fluorescence with polarization-sensitive modalities.
n
n
n
If, after alignment, images still show unexpected background or weak signal, cross-reference the troubleshooting guidance in Avoiding Bleed-Through, Autofluorescence, and Background, and revisit Matching Filters to Fluorophores to confirm your spectral selections.
nn
Avoiding Bleed-Through, Autofluorescence, and Background
n
In fluorescence microscopy, the signal of interest is often orders of magnitude dimmer than the excitation light. Controlling unwanted light is therefore central to achieving crisp, quantitative images. The three most common culprits are spectral bleed-through between channels, autofluorescence from materials, and scattered or stray light. This section gives a structured approach to reducing each, with cross-links to relevant design choices in Filter Technology and Multicolor Imaging Strategies.
n
Reduce spectral bleed-through
n
- n
- Tighten passbands where necessary. Switching from broad to narrower excitation and emission filters in the affected channels often reduces crosstalk.
- Use single-band filters and sequential acquisition when two fluorophores have substantial spectral overlap. Image one channel at a time with the corresponding excitation and emission filters, then switch to the next channel. This simple mechanical or electronic sequence can dramatically improve separation.
- Adjust dichroic cutoffs if the transition region overlaps the emission band you wish to detect. A dichroic with a steeper slope and a slightly shifted cutoff may provide the necessary separation.
- Balance exposure and intensity. Overpowering one channel’s excitation can cause scattered light or tail leakage into another channel. Reducing intensity or exposure for the dominant channel can help.
n
n
n
n
n
Minimize autofluorescence
n
- n
- Choose low-autofluorescence materials for slides, coverslips, and mounting media when possible. Some glasses and adhesives fluoresce under blue or UV illumination, adding unwanted background.
- Shift excitation if practical. If a particular excitation band drives strong background from the mounting medium or substrate, consider selecting a slightly different band with acceptable excitation efficiency (see Matching Filters to Fluorophores for selecting among multiple excitation lobes).
- Use appropriate emission filters to exclude wavelengths where the substrate emits strongly.
n
n
n
n
Control scattered light and reflections
n
- n
- Inspect and clean optical surfaces, especially the dichroic and emission filter. Fingerprints and dust scatter light efficiently.
- Check for internal reflections. Flat, parallel surfaces can form unwanted etalons. If you suspect ghost images, consider small tilts (within manufacturer guidance) or ensure anti-reflective coatings are present on neighboring optics.
- Block unused ports and stray paths. Open ports can admit ambient light that adds background.
n
n
n
n
Finally, monitor your detector settings. Cameras with adjustable gain and offset can exaggerate background if mis-set. Ensure that you are operating within the linear response range for quantitative imaging and that readout modes are appropriate for the signal level.
nn
Multicolor Imaging Strategies: Single-Band, Multiband, and Spectral Unmixing Accessories
n
Imaging two or more labels on the same specimen introduces a new dimension of complexity: channel separation, switching speed, and detector targeting. This section focuses on optical accessories and strategies that address these needs while complementing the filter selection considerations in Matching Filters to Fluorophores.
n
Single-band sets with sequential imaging
n
One robust strategy is to use single-band filter sets for each fluorophore and image the channels sequentially. Advantages include:
n
- n
- Maximum spectral isolation – Each excitation and emission pair is tuned precisely to a single fluorophore.
- Simplicity of interpretation – Lower risk of bleed-through simplifies analysis.
- Flexibility – Easy to add or change channels by swapping cubes or filters.
n
n
n
n
The trade-off is switching time. Mechanically moving cubes or wheels adds latency, though with modern motorized systems this is often manageable for most fixed or moderately dynamic specimens. Electronic source switching (for LEDs or lasers) can be very fast and, combined with motorized filter selection, enables efficient sequential imaging.
n
Multiband filter sets
n
Multiband filters (dual-band, triple-band, etc.) pass multiple excitation and emission bands through a single cube. They are advantageous when you need rapid switching or simultaneous illumination. Considerations include:
n
- n
- Band placement – Ensure the passbands correspond precisely to the fluorophores’ excitation and emission peaks to minimize crosstalk.
- Detector strategy – If imaging multiple bands simultaneously, use appropriate beam-splitting accessories or multi-channel detectors to avoid mixing signals on a single sensor.
- Intensity balance – One fluorophore may be brighter than another. Adjust excitation intensities independently, if possible, to maintain dynamic range without saturating.
n
n
n
n
Multiband sets are powerful for fast imaging workflows but require careful validation. If performance is borderline, reverting to single-band sequential acquisition can often rescue a challenging dataset.
n
Spectral detection and unmixing
n
Some systems use spectral detection, capturing a range of emission wavelengths and then separating fluorophore contributions computationally. While this approach is beyond the scope of basic accessories, note the optical prerequisites:
n
- n
- Broad and clean emission collection – Emission filters might be longpass or relatively broad bandpass to transmit the range of interest without strong ripples or residual excitation leakage.
- Stable illumination – For accurate unmixing, illumination stability across acquisitions is helpful.
- Reference spectra – Accurate reference spectra for each fluorophore under your imaging conditions are essential for robust separation.
n
n
n
n
Spectral strategies can relieve some pressure on filter selectivity but do not eliminate the need for high blocking at excitation wavelengths. Optical cleanliness and correct dichroic choice remain essential; see Filter Technology for blocking considerations.
nn
Compatibility, Mechanical Considerations, and Cube Formats
n
Before purchasing or swapping filters and cubes, verify mechanical and optical compatibility with your microscope. While the principles are universal, the physical formats are not. Here are key points to check:
nn
 n
nn
nn
- n
- Cube format – Different microscope models use specific cube housings. Ensure the filter set is built into the correct cube type for your turret. Adapters exist in some cases, but direct compatibility is safest.
- Filter dimensions and thickness – Excitation and emission filters are commonly round or rectangular and come in standard diameters and thicknesses, but dimensions vary by system. Confirm sizes and thickness tolerances before ordering replacements.
- Dichroic substrate size and orientation – Dichroics are typically rectangular mirrors designed for 45° incidence. Ensure the mirror fits the cube’s slot and is oriented per manufacturer guidance.
- Clear aperture – The usable optical aperture of the filters and the cube should not vignette the beam. Check that the clear aperture is adequate for your objectives and camera sensor size.
- Coating durability – For demanding environments, hard-coated filters are generally advantageous due to their resistance to environmental changes and cleaning.
- Environmental conditions – Avoid excessive humidity and temperature swings that could promote condensation or degrade adhesives in accessories neighboring the filters.
n
n
n
n
n
n
n
If you plan to integrate external accessories—such as emission splitters for two-camera setups or add-on beamsplitters—map out the physical stack and confirm that each element has sufficient aperture and is oriented correctly. Also verify that any additional optics do not reintroduce spectral components you worked to eliminate (for example, by adding a beamsplitter with poor blocking near an excitation line). Attention to these mechanical details helps maintain the optical performance described in Matching Filters to Fluorophores and Filter Technology.
nn
Frequently Asked Questions
n
How do I know if my filters are causing excitation leakage?
n
Common symptoms include a bright haze across the image, signal that does not localize to fluorescent structures, or a background that scales directly with excitation intensity even when the sample lacks the corresponding fluorophore. To diagnose, try these steps:
n
- n
- Block the excitation and observe whether the background persists. If it disappears, leakage is likely.
- Swap emission filters with a known high-blocking filter for the excitation wavelength. If background improves significantly, the previous emission filter was insufficiently blocking or misoriented.
- Inspect for reflections from the dichroic or nearby optics. Ghost images can masquerade as leakage.
n
n
n
n
For a deeper dive into blocking specifications and angle effects, review Filter Technology, Optical Density, and Angle Sensitivity.
n
Can I use a single multiband set for all my fluorophores?
n
It depends on the specific fluorophores and the imaging goals. A well-designed multiband set can work efficiently for some combinations, especially when the source and detector support rapid switching or simultaneous acquisition with proper separation. However, if fluorophore spectra are close or if quantitative accuracy is essential, single-band sequential imaging usually provides cleaner separation and greater flexibility. When in doubt, start with single-band sets to validate the channels and only then consider multiband sets to accelerate workflows.
nn
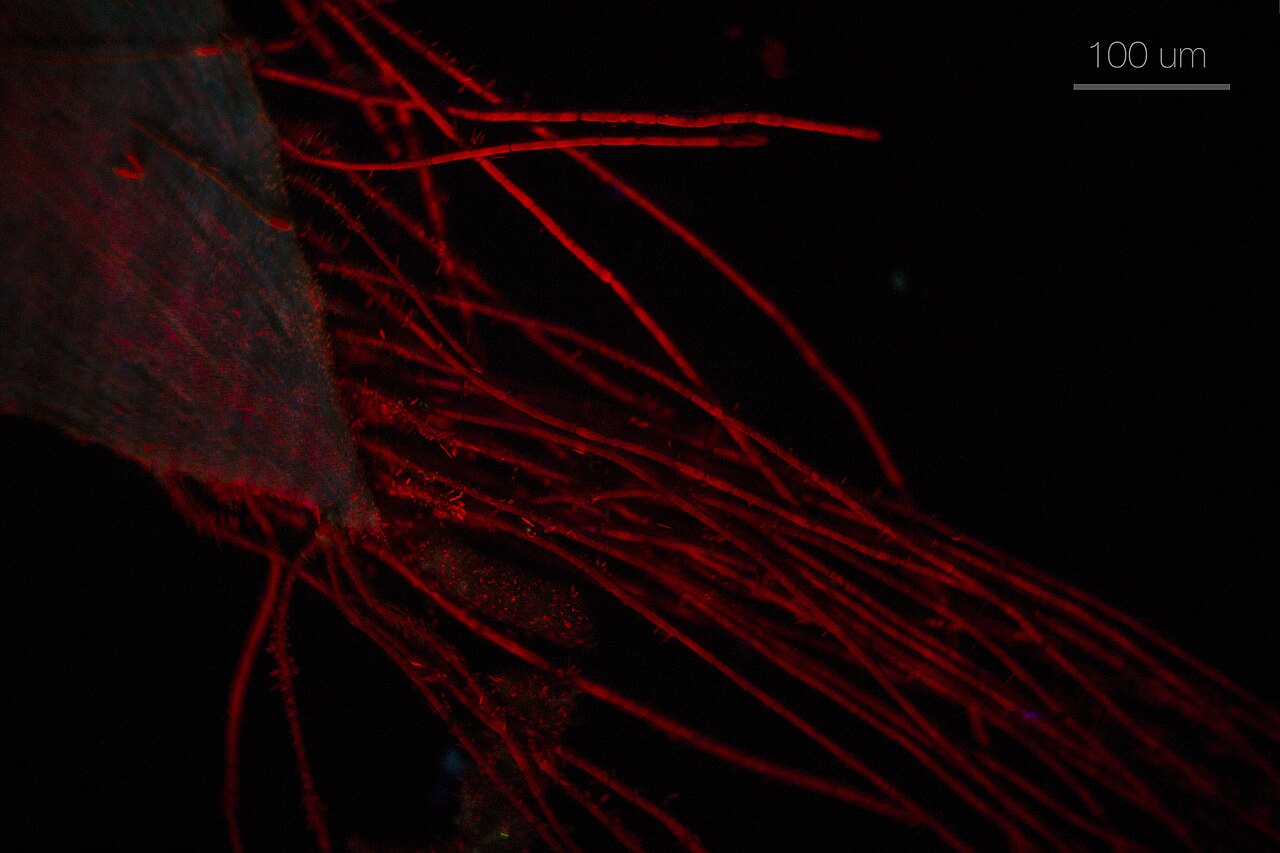
Final Thoughts on Choosing the Right Fluorescence Filters and Light Source
nn
 n
nn
nn
The clarity and reliability of fluorescence images depend on a tightly integrated trio: well-chosen filters, a matching light source, and an aligned, clean optical path. Start with your fluorophores’ spectra, choose excitation and emission bands that maximize desired signal while minimizing background, and pick a dichroic with a steep, well-placed cutoff. Align the choice with your illumination—LEDs for simplicity and stability, lasers for precision and intensity, and arc/metal-halide sources where broadband power or legacy systems fit the need. Then, protect that investment through careful setup, good handling, and routine checks for cleanliness and stray light.
n
If you found this guide helpful, explore related topics in our series on microscope accessories and consider subscribing to our newsletter. We share practical, technically accurate articles each week to help you get the most from your microscope—whether you’re a student, educator, or enthusiast advancing your imaging skills.



